Introduction: The Ongoing Battle Against Prostate Cancer
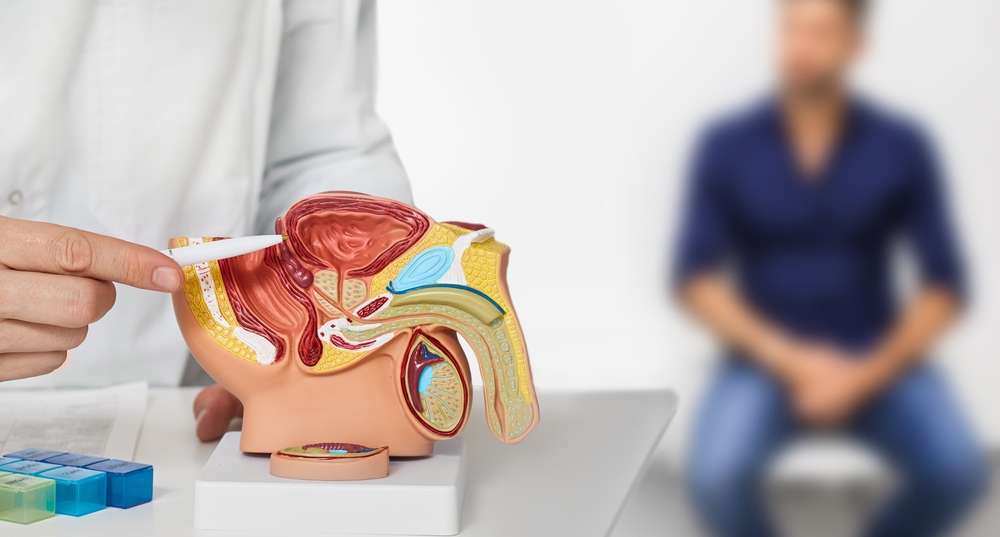
Prostate cancer, a formidable adversary and the most prevalent cancer among men (excluding skin cancer), persists as a significant global health challenge. With over 288,000 new cases diagnosed annually in the United States alone, advancements in addressing the complexities of advanced and metastatic stages are crucial. In a recent breakthrough, a comprehensive study featured in Science Advances delves into the intricate role of the enzyme SMYD3, shedding new light on its involvement in the progression of prostate cancer to more dangerous and aggressive forms.
Probing the Enigmatic Role of SMYD3
The intricate journey of unraveling the enigmatic role of SMYD3 in cancer has been a persistent and intriguing pursuit within the scientific community. This protein, with its fascinatingly heightened presence in cancerous tumors when contrasted with its levels in healthy tissue, has spurred an array of investigations aiming to decipher its implications in the realm of cancer biology. Erin Green, an esteemed Associate Professor of Biological Sciences at the University of Maryland, Baltimore County (UMBC), stands as the senior author spearheading one of these comprehensive studies.
The pursuit of understanding SMYD3’s involvement in cancer is fraught with complexities and occasional contradictions, as elucidated by the existing literature. The intricate web of information surrounding this protein has posed challenges for researchers attempting to unveil its role in the intricate dance of cellular processes leading to cancer development and progression.
SMYD3, a member of the SET and MYND domain-containing protein family, has been recognized for its histone methyltransferase activity, implying its involvement in the epigenetic regulation of gene expression. The allure of its heightened expression in cancerous tissues lies in the potential it holds as a therapeutic target or a diagnostic marker. However, the path to understanding SMYD3’s exact role is riddled with nuances that necessitate a careful dissection of its various interactions within the cellular milieu.
Erin Green’s study, positioned at the forefront of this scientific exploration, seeks to shed light on the intricate mechanisms by which SMYD3 influences cancer biology. Green’s emphasis on the persistent interest in deciphering the mysteries of this protein underscores the urgency and significance attributed to understanding its role in the complex tapestry of cancer-related processes.
The existing literature serves as a treasure trove of information and, at times, a labyrinth of contradictions. The multifaceted nature of SMYD3’s functions, its interactions with other cellular components, and its implications in diverse cancer types contribute to the intricate narrative that scientists strive to unravel. The complexities inherent in this quest necessitate a comprehensive and nuanced approach that goes beyond surface-level observations.
As researchers delve deeper into the molecular intricacies of SMYD3, they grapple with the challenge of reconciling seemingly conflicting data and deciphering the subtle nuances that govern its role in cancer. The journey to unraveling SMYD3’s mysteries extends beyond laboratory experiments, encompassing bioinformatics analyses, clinical correlations, and a continual refinement of experimental methodologies to capture the elusive details that define its contributions to cancer pathogenesis.
In conclusion, the quest to decipher SMYD3’s role in cancer is a captivating scientific journey marked by heightened curiosity and the persistent pursuit of understanding. Erin Green’s study, situated within this dynamic landscape, reflects the dedication of researchers to unravel the complexities surrounding SMYD3, even in the face of a literature that presents a complex and sometimes contradictory picture. As the scientific community collaborates and builds upon existing knowledge, the hope is that the intricate puzzle of SMYD3’s involvement in cancer will gradually yield its secrets, offering new avenues for therapeutic interventions and diagnostic advancements in the ongoing battle against cancer.
The Evolution of Understanding
Early studies proposed that SMYD3 operated within a cell’s nucleus, exerting influence by directly modifying DNA and impacting gene expression. However, Nicolas Reynoird, a co-author of the current study, challenged this conventional wisdom. In a pivotal 2014 paper, Reynoird and collaborators suggested an alternative mechanism, proposing that SMYD3 worked outside the nucleus, activating MAP kinases known for their role in promoting tumor growth.
Insights from Sabeen Ikram’s Key Experiments
Led by postdoctoral fellow Sabeen Ikram at Stanford University, the recent experiments built upon Reynoird’s work, providing more clarity on how SMYD3 potentially triggers metastatic prostate cancer through the MAP kinase signaling pathway. The study serves as a bridge, connecting the dots between the abundance of SMYD3 and the heightened activation of MAP kinase, rekindling interest in SMYD3 as a potential therapeutic target.
Deeper Insights into the Mechanisms of Metastasis
Ikram’s experiments, conducted both in cultured cells and in mice, revealed that SMYD3’s likely role involves the addition of methyl groups to MAP kinase, propelling the metastatic process. Notably, the inactivation of SMYD3 significantly reduced the likelihood of metastasis. Encouragingly, existing SMYD3 inhibitors demonstrated efficacy in killing cancer cells in laboratory settings, opening avenues for potential therapeutic applications.

Beyond Metastasis: Vimentin and the Unveiling of a Positive Feedback Loop
Ikram’s findings went beyond metastasis, shedding light on SMYD3’s impact on vimentin, a well-established marker of cancer progression. Intriguingly, SMYD3’s influence was specific to vimentin within a broader group of similar proteins. Additionally, the study unveiled SMYD3’s creation of a positive feedback loop within cells, contributing to the sustained overabundance of this critical enzyme.
Future Avenues and the Therapeutic Horizon
While the study primarily focused on prostate cancer, Green underscores the need to explore whether similar mechanisms operate in other cancer cell types. The potential utilization of SMYD3 as a therapeutic target offers promising avenues, especially considering the availability of existing SMYD3 inhibitors. Green envisions a broader exploration of these inhibitors, suggesting untapped potential in drug repurposing for enhanced cancer treatment strategies.
Conclusion: Illuminating the Path Forward
In the vast landscape of medical research, the profound insights derived from the study on SMYD3’s intricate role in prostate cancer progression mark a significant milestone. Prostate cancer, standing as one of the most prevalent cancers affecting men globally, has long posed a formidable challenge to researchers and healthcare professionals alike. The sheer number of new cases diagnosed annually in the United States, exceeding 288,000, underscores the urgency for breakthroughs in addressing the complexities of advanced and metastatic stages.
The study, featured in Science Advances, has not only expanded our understanding of the role played by the enzyme SMYD3 but has also illuminated potential avenues for therapeutic interventions. The elevated presence of SMYD3 in cancerous tumors compared to healthy tissue has been a subject of intrigue and scrutiny. Erin Green, the senior author of the study and Associate Professor of Biological Sciences at the University of Maryland, Baltimore County (UMBC), emphasizes the persistent interest in unraveling the mysteries surrounding this protein.
The journey of comprehending SMYD3’s involvement in cancer progression has seen an evolution in understanding. Early studies suggested that SMYD3 operated within a cell’s nucleus, directly modifying DNA and impacting gene expression. However, this conventional wisdom faced a challenge from Nicolas Reynoird and collaborators in a pivotal 2014 paper, proposing an alternative mechanism. They suggested that SMYD3 worked outside the nucleus, activating MAP kinases known for their role in promoting tumor growth.